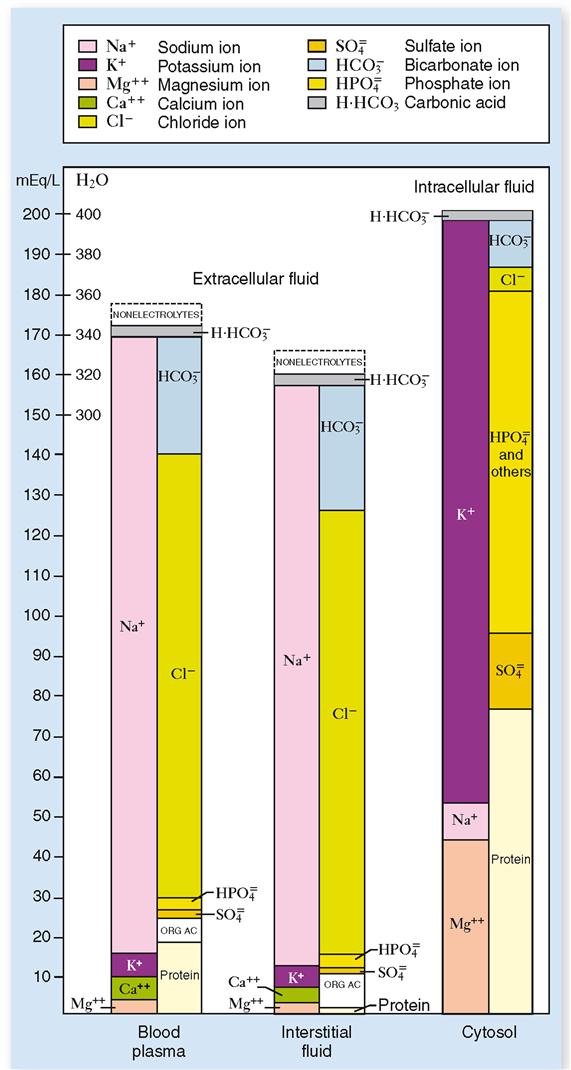
Electrolyte composition of body fluid compartments

As previously discussed, circulating ADH increases water reabsorption from the collecting tubules with a subsequent increase in U osm and a decrease in urine volume. Above this level, a progressive and relatively linear rise occurs in ADH secretion from the posterior pituitary. Normally, the osmotic threshold for ADH release is reached as serum osmolarity exceeds the mid-280 mOsm/L range. Osmoreceptors in the hypothalamus control thirst and the secretion of antidiuretic hormone (ADH). 9.4) resulting in no more than a 1% to 2% normal variation in plasma osmolality, despite wide fluctuations in water and solute intake. Sodium concentration is maintained by an intricate balance of water intake and solute excretion ( Fig. Thus, a normal serum sodium of 140 mEq/L contributes 280 mOsm/L: 140 mOsm from the serum sodium and a further 140 mOsm from the anions electrically balancing the sodium. Hence, in healthy individuals, the major determinant of plasma osmolality becomes the sodium concentration and its anionic counterparts, as estimated by doubling the sodium concentration. Sodium is balanced by plasma cations, primarily chloride and bicarbonate, to maintain electroneutrality. In the absence of diabetes mellitus or renal insufficiency, the contributions to osmolality of glucose and BUN normally are quite small (for example, a normal serum glucose of 100 mg/dL contributes 5.6 mOsm/L and a normal serum BUN of 10 mg/dL only 3.6 mOsm/L, for a total of 9.2 mOsm/L to total serum osmolality). Where serum or plasma sodium is measured in mEq/L and serum or plasma glucose and BUN in mg/dL.

Thus, in these patients, the kidneys respond as though they are volume depleted, with increased sodium avidity (low U Na +) despite a pathologically expanded TBW. The end result, then, is total body and extracellular salt and water expansion but no correction of the low ECV. Due to cardiac insufficiency, however, the increased vascular volume still does not effectively perfuse the kidney. Subsequent renal hypoperfusion activates the release of renin and up-regulation of renal sodium and water retention by angiotensin II and aldosterone.

For example, children with congestive heart failure have a decrease in their ECV due to decreased cardiac output. In certain clinical conditions, these homeostatic mechanisms are altered, resulting in an aberrant fluid and electrolyte balance. Thus, the tightly controlled regulation of ECV is normally maintained by a number of factors, including the size of the extracellular space, the relative efficacy of perfusion within the intravascular space, and hormonal regulation of sodium and water excretion by the kidney. Clinically, this movement is manifested by ascites and peripheral edema, as a direct result of interstitial space expansion or what is often termed third-space losses. For example, patients with marked hypoalbuminemia, such as children with nephrotic syndrome, have an increased movement of fluid from the intravascular space into the interstitial space due to decreased intravascular oncotic pressure. Changes in these processes will lead to a redistribution of water within these extracellular compartments.
The relative volumes of the interstitial and intravascular spaces are determined by the balance of these osmolar forces generated by plasma proteins and hydrostatic forces, which tend to move fluid out of the intravascular space and into the interstium. As a result, the osmotic pressure difference between the intravascular and interstitial fluids is due to plasma proteins that cannot traverse the capillary wall and are, thus, able to hold water in the intravascular space. Unlike the cell membrane, the capillary is permeable to sodium, potassium, and glucose. As noted, the extracellular fluid is divided into interstitial fluid and plasma water via the vascular capillary bed.
